|
adsorption (as dew droplets) onto solid surface at pressures and temperatures higher than the species' triple point-also reversible as evaporation.absorption into the surface of a liquid (either of the same substance or one of its solvents)-is reversible as evaporation. 
In clouds, this can be catalyzed by water-nucleating proteins, produced by atmospheric microbes, which are capable of binding gaseous or liquid water molecules.Ī few distinct reversibility scenarios emerge here with respect to the nature of the surface. When the transition happens from the gaseous phase into the solid phase directly, the change is called deposition.Ĭondensation is initiated by the formation of atomic/molecular clusters of that species within its gaseous volume-like rain drop or snow flake formation within clouds-or at the contact between such gaseous phase and a liquid or solid surface. It can also be defined as the change in the state of water vapor to liquid water when in contact with a liquid or solid surface or cloud condensation nuclei within the atmosphere. The word most often refers to the water cycle. Condensation forming in the low pressure zone above the wing of an aircraft due to adiabatic expansionĬondensation is the change of the state of matter from the gas phase into the liquid phase, and is the reverse of vaporization. 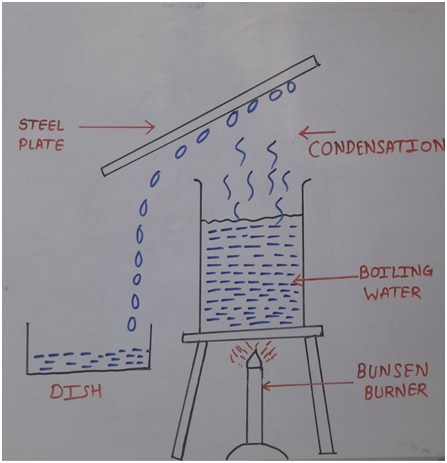
Conductivity increases with airflow in an open evaporator.For other uses, see Condensation (disambiguation). Between the evaporator and the outside air, the medium conductivity is also proportional. The difference between the pressure of the water vapour in the free air and the vapour near the saturated evaporative material directly correlates to the rate of spread or evaporation.Įvaporation is proportional to the saturation deficit when the liquid and the air are both at the same temperature. Diffusion is investigated, but it is the process through which liquid water changes into a gaseous state. The air becomes more humid due to evaporation, particularly from the sea and the soil. Read More: Polar Vortex Evaporation and Humidity A special case of evaporation is transpiration which entails the loss of water from the leaves and stems of the plants. The temperature at which the water starts evaporating is the latent heat of vaporization. There is more evaporation from the ocean than from the land. Evaporation is faster in dry air than in wet air. The amount and rate of evaporation at a particular place depend upon the aridity, temperature, and air movement. It is how liquid water changes into water vapour on heating. As a result, evaporation grows in direct proportion to air movement. The unsaturated layer replaces the saturated layer as a result of air movement. The concentration of air in the parcel and the absorption of water both increase as the temperature rises. The temperature at which water vaporises is referred to as the latent heat of vaporisation. 
The process of evaporation turns water from a liquid to a gas. Evaporation DefinitionĮvaporation is mostly caused by temperature. This article discusses Evaporation and Condensation which is useful for competitive exams such as the UPSC exam. Condensation is a phase change that occurs regardless of temperature. Before reaching its boiling point, a liquid evaporates. Condensation is the inverse process that converts water vapour to tiny droplets of water. UPPSC RO ARO Previous Year Question PapersĮvaporation is the process by which water transforms into a vapour.UPSC Geo Scientist Eligibility Criteria. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
